JEE Mains 2026 21st January Shift 2
Chemistry
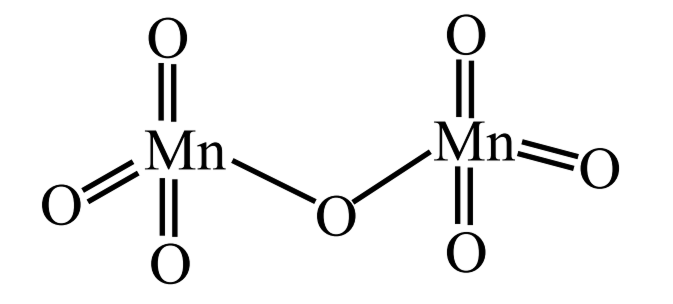
Given below are some of the statements about Mn and . Identify the correct statements
A. Mn forms the oxide in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn
C. is an ionic oxide.
D. The structure of consists of one bridged oxygen.
Choose the correct answer from the options given below:
A
A, B, C and D
B
A, B and D Only
C
A, C and D Only
D
A, B and C Only
Select an option to instantly check whether it is correct or wrong.