Exam 2026 21st January Shift 1
Chemistry
For the reaction, , graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is
B. As in graph is positive, will not dissociate into at all.
C. Reverse reaction will go to completion.
D. When 1 mole of changes into equilibrium mixture, value of
E. When 2 mole of , changes into equilibrium mixture, for equilibrium mixture is
E. When 2 mole of , changes into equilibrium mixture, for equilibrium mixture is 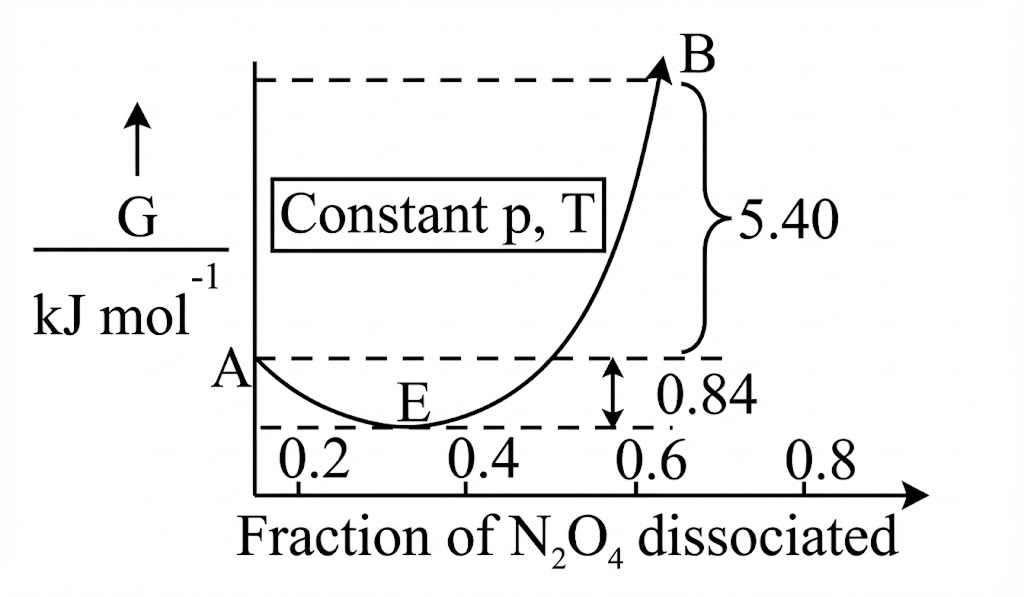 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :
 Choose the correct answer from the options given below :
Choose the correct answer from the options given below :A
D and E only
B
C and E only
C
A and D only
D
B and C only
Select an option to instantly check whether it is correct or wrong.




